lorem ipsum
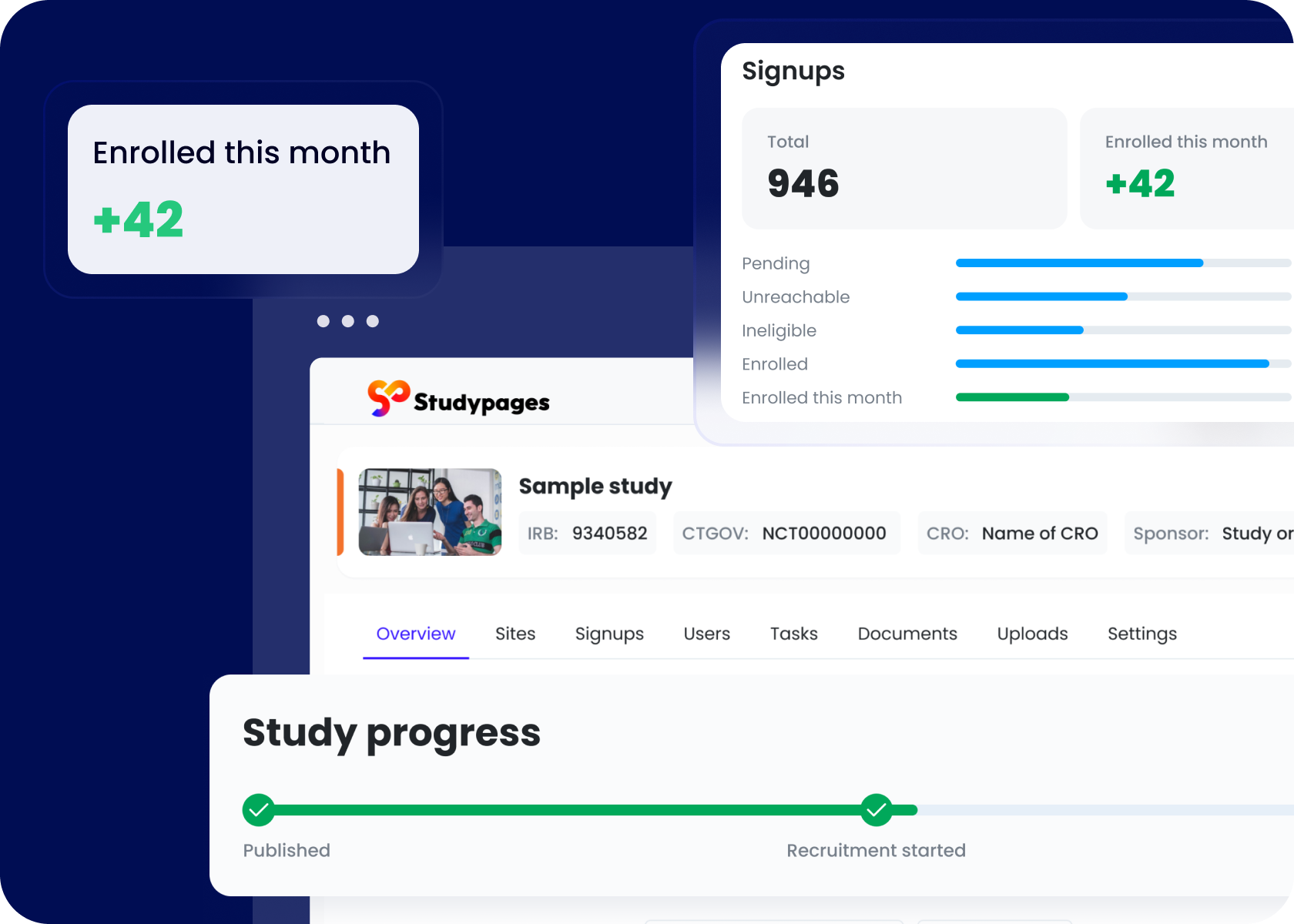
Workflows to enroll end execute trials efficiently
Turn your research site into a powerhouse of efficiency and community engagement. Studypages provides study teams with tools to connect and recruit study participants, integrate effortlessly with trial technologies, and enhance productivity.

Integrate systems, data,
and people
Lorem ipsum dolor sit amet consectetur. Pharetra mi amet est donec nibh interdum vitae sit. Lorem ipsum dolor sit amet consectetur. Pharetra mi amet est donec nibh interdum vitae sit.

Manage study content and outreach efforts
Lorem ipsum dolor sit amet consectetur. Sed vestibulum sed ipsum volutpat mauris ultricies. Eu vitae odio proin leo arcu.

Enhance study teams’ ability to recruit and run studies
Lorem ipsum dolor sit amet consectetur. Urna mauris tortor facilisis hendrerit amet ornare sed orci. Egestas fringilla nibh. Lorem ipsum dolor sit amet consectetur. Urna mauris tortor facilisis hendrerit amet ornare sed orci. Egestas fringilla nibh.









.jpg)


